High-throughput sequencing systems routinely generate large volumes of data with strong internal quality metrics, yet these metrics primarily reflect base-calling confidence rather than overall analytical validity. In practice, most sequencing errors originate upstream of detection or arise from instability in supporting equipment and workflow execution. While chemistry and bioinformatics are often emphasised, reproducibility in sequencing is heavily dependent on mechanical precision, environmental control, and consistency of instrumentation (Schirmer et al., 2015; Fox et al., 2014).
A primary source of variability lies in sample and library preparation, where small deviations in liquid handling propagate into measurable bias. Manual pipetting introduces volumetric error and operator-dependent variability, particularly at low volumes typical of next-generation sequencing workflows. Even minor inconsistencies in reagent ratios affect adapter ligation efficiency, amplification kinetics, and fragment representation. Studies have demonstrated that library preparation is a dominant contributor to inter-sample variability, often exceeding platform-level error (Aird et al., 2011). Automated liquid handling systems reduce coefficient of variation and improve reproducibility across batches, particularly in high-throughput or regulated environments where consistency is critical.
Thermal control during amplification is another key determinant of data quality. PCR bias is influenced not only by enzyme fidelity and cycle number, but also by thermal uniformity and ramp rate accuracy. Poorly calibrated or ageing thermal cyclers can introduce well-to-well variation, leading to uneven amplification and reduced library complexity. This manifests as coverage bias and preferential enrichment of certain fragments. Research into PCR amplification bias has shown strong dependence on thermal cycling conditions, particularly in GC-rich regions (Aird et al., 2011). High-quality thermal cyclers with validated block uniformity significantly reduce this source of variability.
Fragment size distribution directly impacts sequencing efficiency and data interpretation. Inaccurate sizing leads to incorrect molarity calculations, affecting cluster density and read quality. Overestimation of fragment size results in underloading and reduced output, while underestimation contributes to overclustering and signal interference. Automated electrophoretic systems provide higher resolution and reproducibility compared to gel-based methods, enabling more accurate normalisation across samples. This is particularly important in multiplexed workflows where consistent fragment distribution is required to minimise bias (van Dijk et al., 2014).
Quantification methods further contribute to variability. Spectrophotometric approaches are susceptible to interference from contaminants such as proteins, salts, and free nucleotides, often resulting in overestimation of DNA concentration. This directly impacts loading efficiency and sequencing performance. Fluorescence-based quantification methods, particularly those specific to double-stranded DNA, provide greater specificity and are widely recommended for library preparation workflows (Simbolo et al., 2013). When combined with accurate fragment sizing, these approaches significantly improve loading consistency and reduce run-to-run variability.
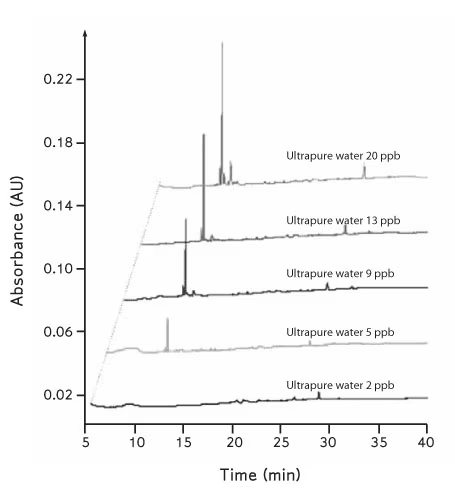
Environmental control is frequently underestimated as a source of sequencing error. Airborne contamination, aerosolised nucleic acids, and surface residues can introduce low-level DNA carryover, particularly in pre-amplification workflows. This is a well-documented issue in both clinical and research sequencing environments, where contamination can lead to false-positive variant detection or spurious taxa in metagenomic datasets (Salter et al., 2014). Physical separation of pre- and post-PCR processes, combined with clean bench environments and dedicated equipment, remains one of the most effective mitigation strategies.
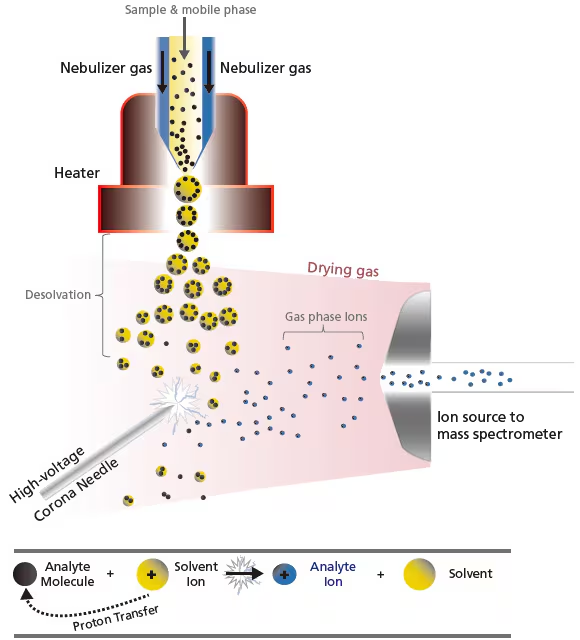
Within sequencing instruments themselves, fluidics and optical systems must operate within narrow tolerances to maintain performance. Variability in reagent delivery, flow cell loading, or imaging can introduce systematic error, although these effects are generally secondary to upstream issues. Platform-specific error profiles, such as substitution errors in short-read systems or indel bias in long-read technologies, are well characterised and can be mitigated through depth and computational correction (Goodwin et al., 2016). However, consistent instrument performance still depends on routine calibration and maintenance of mechanical and optical components.
Index misassignment and cross-contamination during multiplexing further illustrate the importance of physical workflow control. Index hopping and sample cross-talk have been observed in patterned flow cell systems and are exacerbated by residual free adapters and high loading concentrations (Kircher et al., 2012). Thorough library cleanup, accurate pooling, and the use of unique dual indexing strategies reduce these risks and improve data fidelity.
A common limitation in sequencing workflows is the overreliance on instrument-generated quality metrics such as Q30 scores and total read counts. While these metrics provide useful indicators of run performance, they do not capture biases introduced by sample preparation, contamination, or library composition. High-quality scores can therefore coexist with biologically inaccurate or misleading results. This reinforces the need to control physical variables throughout the workflow rather than relying solely on downstream validation (Schirmer et al., 2015).
In practice, the most effective approach to reducing sequencing error is system-wide standardisation supported by reliable instrumentation. Consistent liquid handling, precise thermal control, accurate quantification, and controlled environments collectively reduce variability and improve reproducibility. In high-throughput and regulated settings, these factors are often the primary determinants of data quality.
Ultimately, sequencing accuracy is governed less by the headline performance of the instrument and more by the integrity of the workflow that supports it. Equipment that reduces human variability, maintains tight physical tolerances, and enables consistent execution across samples is central to producing reliable and interpretable sequencing data.